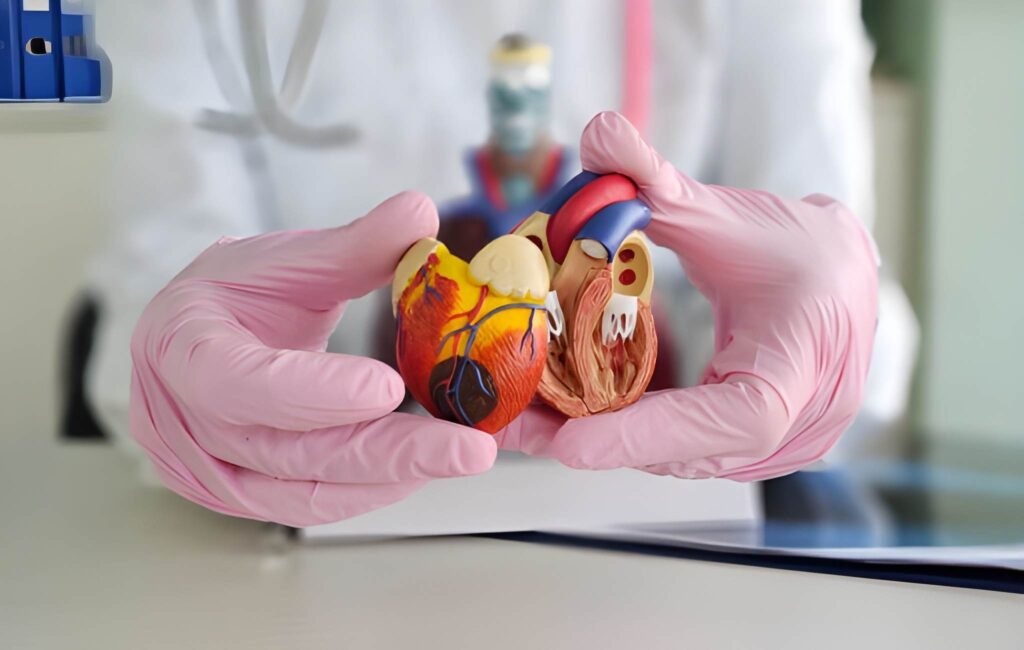
Congenital heart disease remains a significant area of focus for pediatric cardiologists, and perhaps no condition is as classically recognized as Tetralogy of Fallot (ToF). This condition serves as a hallmark of cyanotic heart disease, characterized by a specific cluster of four anatomical abnormalities that permanently alter the way blood flows through the heart and to the lungs. When a newborn arrives with these defects, the balance of their circulatory system is delicate. Without surgical intervention, the infant faces chronic oxygen deprivation, often manifesting as a deep blue tint to the skin. Modern surgery has transformed this once-fatal diagnosis into a manageable condition with high success rates, provided the clinical team understands the intricate interplay of these four structural deviations.
The Four Primary Defects
The term Tetralogy originates from the Greek word for four, reflecting the consistent structural anomalies first described by Etienne-Louis Arthur Fallot in 1888. These defects occur during fetal development, specifically involving the malalignment of the conotruncal septum. This septum is the internal wall that divides the outflow tracts of the heart. When it shifts too far in one direction, it triggers a domino effect of structural changes that define the condition.
Physiology: The Shunt Effect
The core physiological problem in Tetralogy of Fallot is a right-to-left shunt. In a healthy heart, blood from the right side goes exclusively to the lungs. In ToF, because the pulmonary exit is narrowed (stenosis), the blood takes the path of least resistance. It flows through the VSD into the left ventricle and out through the overriding aorta to the rest of the body. This means blood that has never been to the lungs is circulating through the infant's organs. The result is a lower-than-normal concentration of oxygen in the systemic circulation.
The severity of cyanosis is directly proportional to the degree of pulmonary stenosis. If the narrowing is mild, the infant may appear "pink" initially. If the stenosis is severe or complete (pulmonary atresia), the infant will be deeply cyanotic and require immediate medical stabilization via a prostaglandin infusion to keep the ductus arteriosus open.
Managing the Acute Tet Spell
Infants with ToF are prone to "Tet spells," or hypercyanotic episodes. These events occur when an infant cries, feeds, or becomes agitated, causing a sudden drop in systemic vascular resistance or an increase in pulmonary resistance. This dramatically increases the right-to-left shunt, plunging the infant into a state of severe hypoxia. Recognising and treating these spells is a vital skill for both parents and clinical staff.
During a Tet spell, the first action is to place the infant in a knee-to-chest position. This physical maneuver increases systemic vascular resistance by kinking the femoral arteries. This forces more blood to stay in the left ventricle, which in turn pushes more blood back across the VSD and through the pulmonary valve toward the lungs.
If physical maneuvers fail, medical intervention is necessary. Oxygen is administered to promote pulmonary vasodilation. Morphine or fentanyl may be used to calm the infant and reduce the respiratory drive. In severe cases, phenylephrine is used to raise systemic blood pressure and reverse the shunt direction.
Palliative vs. Total Repair
The treatment for Tetralogy of Fallot is strictly surgical. In the past, many infants underwent a staged approach, but today, most centers prefer a primary total repair within the first six months of life. However, certain anatomical variations or very small infants may still require a palliative procedure first to ensure they survive long enough for a permanent fix.
| Procedure | Mechanism | Clinical Indication |
|---|---|---|
| Blalock-Taussig (BT) Shunt | Palliative: Connects subclavian artery to pulmonary artery. | Small infants or those with severe pulmonary atresia. |
| VSD Closure | Total Repair: Patching the hole with synthetic material. | Standard corrective surgery; redirecting left ventricular flow. |
| Pulmonary Valvulotomy | Total Repair: Widening the narrowed pulmonary outflow. | Standard corrective surgery; ensuring blood reaches the lungs. |
| Transannular Patch | Total Repair: Expanding the pulmonary valve ring. | When simple valvulotomy is insufficient for adequate flow. |
Imaging and the Boot-Shaped Heart
Diagnosis typically begins with an echocardiogram, which provides a real-time view of the four defects. However, a chest X-ray often reveals a classic sign known as the "Coeur en Sabot" or boot-shaped heart. This shape results from the right ventricular hypertrophy pushing the apex of the heart upward, while the narrowed pulmonary artery creates a "concave" appearance where the pulmonary segment should normally bulge.
Calculations: Oxygen Saturation
In a healthy infant at sea level, oxygen saturation (SpO2) should remain between 95% and 100%. In an infant with ToF, we manage for "stable hypoxia." We want the SpO2 to be high enough to support growth but we accept that it will be lower than an unaffected peer. Understanding the mixture of blood allows clinicians to predict the infant's tolerance for activity.
Example for a stable ToF infant:
Oxygenated Blood (from lungs): 98%
Deoxygenated Blood (from body): 65%
If the shunt is 1:1 (equal amounts of blood mixing):
(98 + 65) / 2 = 81.5%
Target Saturation: Most ToF infants are considered stable if their resting SpO2 remains between 75% and 85%. If it drops below 70%, surgical urgency increases.
Life After Cardiac Reconstruction
The success of ToF repair is a triumph of modern medicine. Most children who undergo total repair lead active, fulfilling lives. However, they are not "cured" in the traditional sense; they require lifelong follow-up with a congenital cardiologist. The most common long-term issue is pulmonary regurgitation. Because the surgeon often has to widen the pulmonary valve, the valve may leak later in life, eventually requiring a replacement (such as a Melody Valve) during adolescence or early adulthood.
As we move through , advancements in 3D printing and robotic-assisted surgery are allowing for even more precise repairs tailored to the specific anatomical nuances of each infant. By identifying the four defects early and intervening with a methodical surgical plan, we ensure that the "blue baby" of yesterday becomes the thriving, active child of tomorrow. The continuum of care—from prenatal diagnosis to adult congenital follow-up—remains the gold standard for managing the complex architecture of Tetralogy of Fallot.