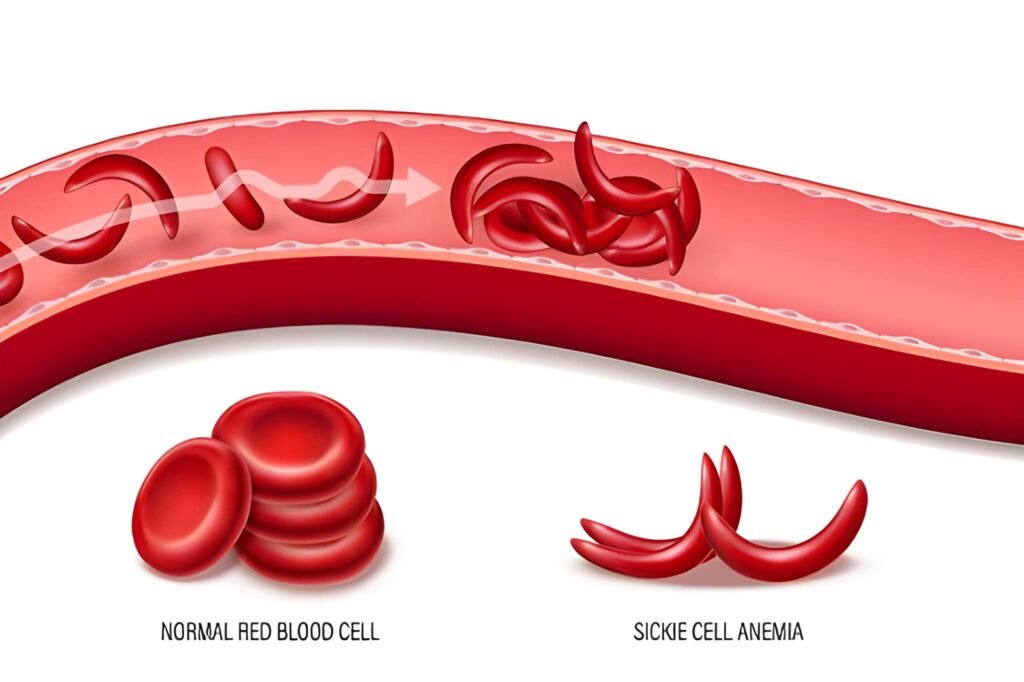
A newborn diagnosed with Sickle Cell Anemia (SCA) starts a life journey that requires meticulous clinical oversight and profound family resilience. Sickle Cell Anemia is a genetic disorder affecting hemoglobin, the protein in red blood cells that carries oxygen throughout the body. In a healthy infant, red blood cells are flexible and round, moving with ease through even the narrowest capillaries. In an infant with SCA, the hemoglobin molecules (Hgb S) stick together when they release oxygen, causing the cells to stiffen and twist into a crescent or "sickle" shape. This structural change triggers a cascade of complications, from localized pain to systemic organ damage, making early diagnosis and prophylactic care the cornerstones of modern neonatal management.
The Mechanics of the Sickle Shape
The core of the disorder lies in a single point mutation in the beta-globin gene. This mutation causes the red blood cells to lose their elasticity. When oxygen levels drop, the hemoglobin S polymerizes into long, rigid fibers. These fibers distort the cell membrane into the characteristic sickle shape. Unlike healthy red blood cells, which live for roughly 120 days, sickled cells survive for only 10 to 20 days. This rapid destruction leads to a state of chronic anemia. Furthermore, the rigid shape makes the cells "sticky," causing them to cluster and obstruct blood flow in small vessels. This obstruction, known as vaso-occlusion, deprives tissues of oxygen and causes intense pain.
The sickle shape does more than just cause pain. Because the body destroys these cells so rapidly, the bone marrow must work at a high velocity to produce replacements. This places a metabolic strain on the infant. Additionally, the spleen, which filters the blood, becomes overworked as it tries to remove the damaged cells. Over time, the spleen sustains repeated micro-injuries, eventually losing its ability to function—a condition known as autosplenectomy. This loss of splenic function significantly increases the infant's vulnerability to life-threatening bacterial infections.
Screening Protocols in the United States
In the United States, newborn screening for Sickle Cell Anemia is a universal mandate across all 50 states. This screening occurs shortly after birth, usually via a heel-prick blood test. The primary goal is the identification of Hemoglobin SS (the disease) or Hemoglobin AS (the trait) before the infant leaves the hospital. Early identification allows the medical team to initiate prophylactic treatments before the first symptoms even appear, dramatically reducing the mortality rate in the first five years of life.
The Genetic Lottery
SCA is an autosomal recessive disorder. This means an infant must inherit two copies of the sickle cell gene—one from each parent—to have the disease. If an infant inherits only one copy, they possess the Sickle Cell Trait (SCT). While individuals with the trait typically lead healthy lives, they can pass the gene to their children. Genetic counseling serves as a vital resource for families following a newborn diagnosis, providing clarity on the risks for future siblings and the broader family tree.
The Protective Window of Hgb F
A remarkable biological phenomenon protects newborns with SCA during their first few months of life. While in the womb, a fetus produces Fetal Hemoglobin (Hgb F). This type of hemoglobin has a much higher affinity for oxygen than adult hemoglobin. Most importantly, Hgb F does not sickle. At birth, Hgb F makes up roughly 60% to 80% of the infant's total hemoglobin. This high concentration acts as a natural shield, preventing sickling and keeping the red blood cells flexible.
The Penicillin Shield and Vaccinations
Because the spleen loses function early in infants with SCA, they face a high risk of infection from encapsulated bacteria, particularly Streptococcus pneumoniae. Before the 1980s, infection was the leading cause of death for these children. Today, clinical protocols mandate the initiation of daily prophylactic penicillin by two months of age. This "penicillin shield" provides a constant baseline of protection while the infant's immune system matures.
| Intervention | Standard Timeline | Clinical Goal |
|---|---|---|
| Penicillin VK | Started by 2 months | Prevention of S. pneumoniae sepsis |
| PCV13 Vaccine | 2, 4, 6, and 12 months | Immunization against pneumococcal strains |
| H. influenzae (Hib) | 2, 4, 6 months | Prevention of bacterial meningitis |
| Folic Acid | Daily supplement | Support for rapid red blood cell production |
Identifying Early Vaso-Occlusive Signs
Parents must become experts in identifying the subtle signs of a vaso-occlusive crisis. In infants, one of the earliest manifestations is Dactylitis, also known as Hand-Foot Syndrome. This involves painful swelling of the hands and feet caused by sickled cells blocking blood flow to the small bones. While dactylitis itself is not life-threatening, it indicates that sickling is actively occurring and requires immediate attention to prevent progression to more severe complications.
Splenic Sequestration: This occurs when a large volume of blood suddenly becomes trapped in the spleen. Symptoms include a rapid drop in hemoglobin, extreme lethargy, and a visibly distended abdomen. This is a medical emergency.
Acute Chest Syndrome: Similar to pneumonia, this involves sickling in the lungs. Watch for rapid breathing, chest pain, and a persistent cough. Immediate oxygen and antibiotics are required.
Fever: Any fever over 101.3 degrees Fahrenheit (38.5 degrees Celsius) in a child with SCA requires immediate hospital evaluation, as it could signal a rapid-onset bacterial infection.
Calculating Fluid Maintenance Needs
Hydration serves as the simplest and most effective way to prevent red blood cells from sickling. Dehydration increases the concentration of hemoglobin within the cell, making sickling more likely. In infants with SCA, we aim for a "high-maintenance" fluid intake. Clinicians use weight-based formulas to determine the baseline volume needed to keep the blood moving and the kidneys functioning efficiently.
Standard Rule for Daily Fluid Volume:
100ml per kg for the first 10kg of body weight.
Example for a 6kg newborn:
6kg * 100ml = 600ml per 24 hours.
Clinical Note: For infants with SCA, providers often recommend increasing this by 20% to 50% during periods of high heat, fever, or physical stress to provide a safety margin against sickling.
Socioeconomic Support and Family Care
Managing SCA in the United States involves navigating a complex healthcare system. Families require access to pediatric hematologists, social workers, and financial resources to manage the lifelong costs of care. The Sickle Cell Disease Association of America (SCDAA) provides essential advocacy and support, helping families connect with clinics and stay informed about emerging therapies like L-glutamine and gene-editing trials.
Equally important is the emotional health of the family unit. The chronic nature of the disease, with its unpredictable crises, can lead to significant caregiver stress. Support groups offer a space for parents to share management strategies and cope with the emotional toll of the diagnosis. When families feel empowered and have a robust medical "home," children with SCA achieve much better long-term health outcomes, proving that the medical management of the disease is as much about the community as it is about the hemoglobin.
The Future of Sickle Cell Care
As we move through , the outlook for newborns with Sickle Cell Anemia is the brightest it has ever been. We are witnessing a revolution in treatment, from the widespread use of Hydroxyurea to the approval of curative gene-editing therapies like Casgevy. While the diagnosis remains serious, the combination of early screening, daily penicillin, and advanced genomic medicine means that today’s newborns can look forward to active, productive lives. By protecting the heritage of the child and the health of the hemoglobin, we ensure that every infant with SCA has the opportunity to thrive.