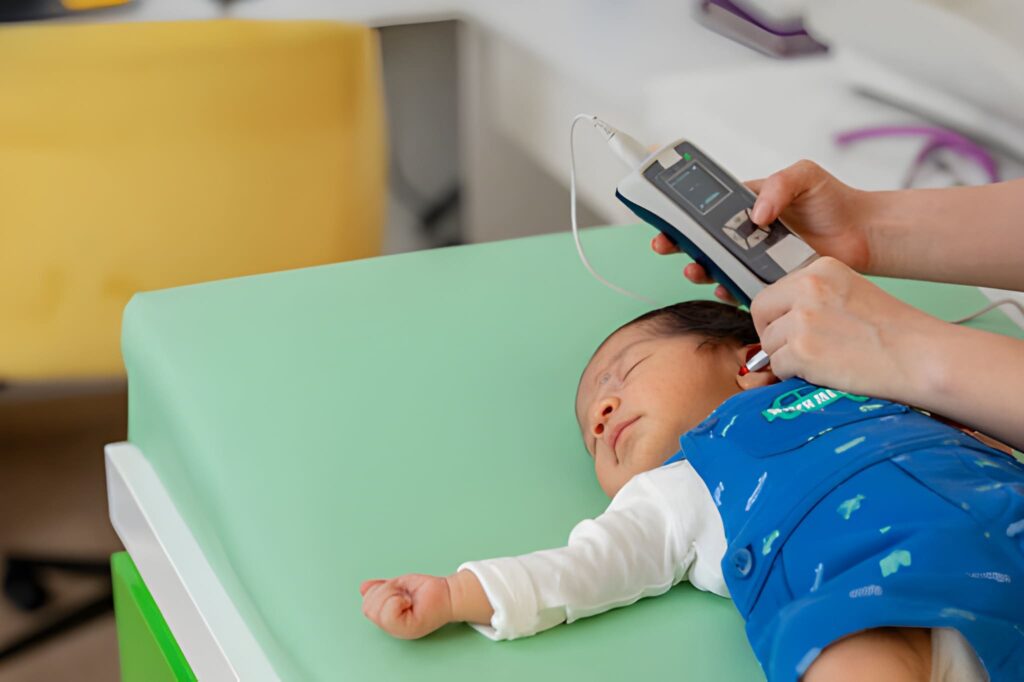
A newborn infant arriving with craniofacial abnormalities (CFA) enters a world where the architectural integrity of the face and the functional pathways of sound are inextricably linked. Craniofacial abnormalities encompass a diverse range of structural deviations, including cleft lip and palate, microtia (small or absent outer ear), and various craniosynostosis syndromes. While the visible structural issues often receive immediate attention, the internal auditory systems are frequently compromised simultaneously. Because the ears, face, and jaw develop from the same embryological structures—primarily the first and second branchial arches—a deviation in one area often signals a deviation in the other. Effective screening for hearing loss in this population requires a departure from standard newborn protocols, demanding a more rigorous, diagnostic-level approach from the very first days of life.
The Link Between Structure and Sound
To understand why infants with craniofacial issues require specialized screening, we must look at embryology. During the first few weeks of gestation, the structures that will become the jaw, the facial bones, and the tiny ossicles of the middle ear begin to take shape. Specifically, the first branchial arch (Mandibular arch) contributes to the formation of the malleus and incus bones, as well as the external ear canal. The second branchial arch (Hyoid arch) contributes to the stapes bone. Any genetic or environmental factor that disrupts the migration of neural crest cells to these arches can result in a spectrum of abnormalities known as oculo-auriculo-vertebral spectrum or hemifacial microsomia.
Infants with a cleft palate, even without other visible ear abnormalities, possess a significantly higher risk for conductive hearing loss. This occurs because the muscles that open the Eustachian tube—the tensor veli palatini—are often misaligned or weakened. This failure leads to chronic fluid accumulation in the middle ear (otitis media with effusion), which acts as a physical barrier to sound waves. In the United States, pediatricians prioritize these infants for early audiological monitoring to prevent speech and language delays.
Standard vs. Specialized Protocols
The Universal Newborn Hearing Screening (UNHS) is the standard of care for healthy newborns in the US. However, for infants with craniofacial abnormalities, the "screening" phase often moves immediately into the "diagnostic" phase. Standard screening typically utilizes a pass/fail model, which may be insufficient for a child with complex anatomy. For instance, an infant with Aural Atresia (absence of the ear canal) cannot undergo a traditional ear-probe test because there is no canal to enter.
The Limitation of Traditional Screening
Traditional screening methods often miss the nuances of a child's specific anatomical barriers. In a healthy newborn, the focus is on identifying any level of hearing loss. In a child with CFA, the focus shifts to identifying the type and degree of loss immediately. Clinical guidelines recommend that these infants skip the initial "screening" and go directly to a comprehensive audiological evaluation by a pediatric audiologist specialized in craniofacial conditions.
Technologies: OAE and AABR Analysis
Two primary technologies dominate the landscape of newborn hearing assessment: Otoacoustic Emissions (OAE) and Automated Auditory Brainstem Response (AABR). While both are valuable, they serve different purposes and possess different limitations in the context of craniofacial deviations.
| Technology | Mechanism | CFA Limitation |
|---|---|---|
| OAE | Measures sound echoes from the cochlea. | Fails if middle ear fluid or canal atresia is present. |
| AABR | Measures brainstem response via electrodes. | Requires skin contact; may be difficult with facial abnormalities. |
| Bone-Conduction ABR | Bypasses the outer/middle ear entirely. | The gold standard for assessing inner ear health in CFA. |
For an infant with microtia or atresia, Bone-Conduction ABR is the critical diagnostic tool. By placing an oscillator on the mastoid bone, clinicians send sound vibrations directly to the cochlea through the skull. This allows the team to determine if the inner ear and nerve are functioning correctly, regardless of the outer ear's structural status. This distinction is vital: if the inner ear is healthy, the child’s hearing potential remains high, even if they require surgical reconstruction of the canal later in childhood.
Associated Syndromes and Auditory Risk
Craniofacial abnormalities often present as part of a larger genetic syndrome. These syndromes carry predictable patterns of hearing loss, allowing the medical team to anticipate the child's needs before the screening even begins. Awareness of these patterns is a cornerstone of neonatal triage.
Characterized by underdevelopment of the cheekbones and jaw. Over 50% of these infants have significant conductive hearing loss due to malformations of the middle ear ossicles (the hammer, anvil, and stirrup). Many also present with microtia and atresia.
Features a small jaw (micrognathia), a tongue that falls back (glossoptosis), and often a cleft palate. The high incidence of middle ear fluid in these infants necessitates aggressive monitoring with tympanometry to ensure fluid does not impede auditory development.
Part of the hemifacial microsomia spectrum, this syndrome often affects one side of the face. Infants frequently have preauricular tags or pits and a high rate of unilateral (one-sided) conductive hearing loss. Because they have one "good" ear, the loss in the affected ear is sometimes overlooked, which can still impact localized sound processing.
The 1-3-6 Rule in Modern Pediatrics
In the United States, the Early Hearing Detection and Intervention (EHDI) program establishes the "1-3-6" framework. While this rule applies to all newborns, it is a strict mandate for those with craniofacial abnormalities, where the risk of delay is compounded by surgical and structural complications.
Statistical Prevalence Calculations
The statistical probability of hearing loss in the craniofacial population is vastly higher than in the general newborn population. While standard newborn hearing loss occurs in roughly 2 to 3 out of every 1,000 births, the numbers for specific CFA categories are significantly more aggressive. Understanding these ratios helps public health officials and hospital administrators allocate the necessary audiological resources.
Cleft Palate Population Prevalence: 90% to 100% (Transient conductive loss due to fluid)
Microtia/Atresia Population Prevalence: 85% to 95% (Persistent conductive loss)
Calculation of Relative Risk (RR):
RR = (Prevalence in CFA group) / (Prevalence in General group)
RR = 90% / 0.3% = 300
Result: An infant with a cleft palate is approximately 300 times more likely to experience hearing impairment than an infant without craniofacial structural issues.
Socioeconomic Factors in Follow-Up Care
The technical success of a hearing screen is meaningless without a robust follow-up system. In the US, socioeconomic context plays a major role in whether a child with CFA receives the long-term care they need. These infants require a "Craniofacial Team," which typically includes a plastic surgeon, otolaryngologist (ENT), audiologist, speech-language pathologist, and geneticist. Coordinating these appointments requires significant parental resources, transportation, and insurance coverage.
Studies show that infants from lower socioeconomic backgrounds are more likely to be "lost to follow-up" after the initial hospital screening. For a child with a structural abnormality, this is particularly dangerous. If middle ear fluid is left untreated, or if a structural blockage is not bypassed with technology early, the child’s brain will miss the "critical period" for language acquisition. This leads to permanent deficits in cognitive and social development that are entirely preventable with modern intervention.
The Path to Auditory Success
As we move through , the focus of neonatal audiology is shifting from passive screening to proactive management. For the newborn with craniofacial abnormalities, the goal is not just to "pass" a test, but to ensure that the auditory brain receives high-quality sound signals despite the structural hurdles. By integrating bone-conduction technology, early Eustachian tube management, and strict adherence to the 1-3-6 guidelines, we can ensure these children achieve communication parity with their peers. The resilient architecture of the human face may be complex, but with a methodical clinical approach, the gift of sound remains accessible to every child, regardless of their starting point.